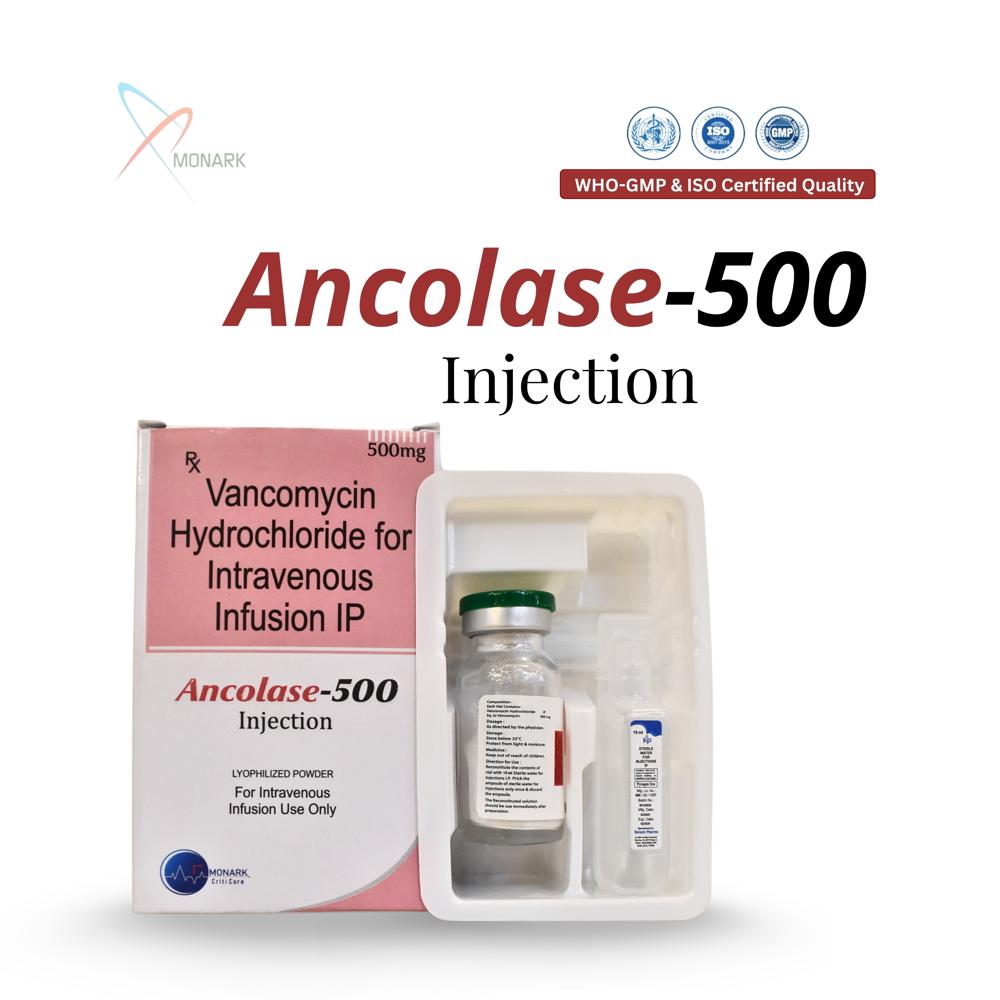
About Vancomycin Hydrochloride 500 MG For Intravenous Infusion IP
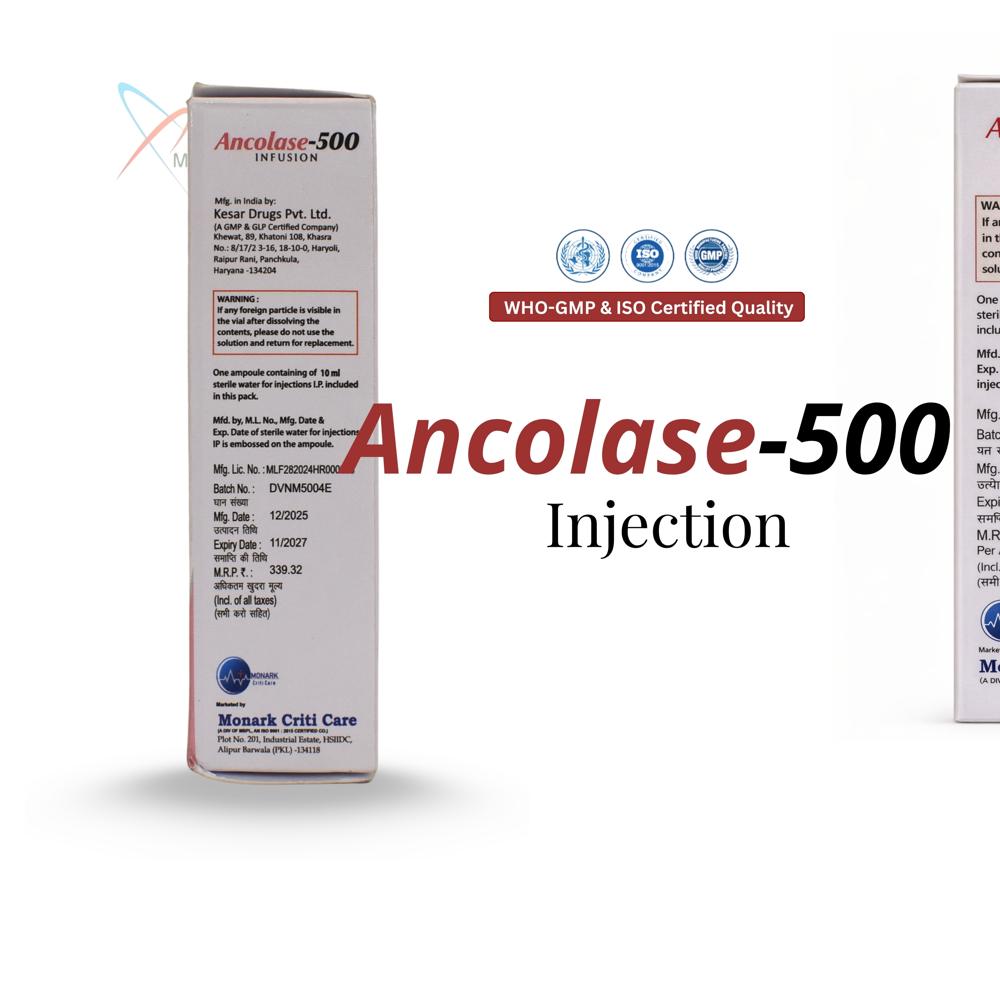
Vancomycin Hydrochloride 500 mg For Intravenous Infusion IP is a lyophilized sterile powder in a single-dose vial containing the equivalent of 500 mg vancomycin base, meeting Indian Pharmacopoeia standards for IV use.
Flexible Dosing for Enhanced Patient CareThe Ancolase-500 formulation allows precise dose adjustments, making it ideal for patients requiring lower infusion volumes. Its flexibility supports personalized therapy, especially in pediatric patients or those with compromised kidney function, while ensuring effective treatment of life-threatening bacterial infections.
Indications and Clinical ApplicationsAncolase-500 is primarily indicated for severe infections caused by susceptible Gram-positive organisms, including MRSA, C. difficile colitis (when administered IV), endocarditis, and bacteremia. Its reliable efficacy and tailored strength enhance its use in step-down therapies and fluid-restricted patients.
Safe Storage and HandlingTo maintain potency, store Ancolase-500 vials in a cool, dry place, away from direct sunlight and moisture. Each vial is meant for single use after reconstitution, ensuring safety and sterility are upheld throughout the administration process.
FAQ's of Vancomycin Hydrochloride 500 MG For Intravenous Infusion IP:
Q: How should Ancolase-500 Vancomycin Hydrochloride be prepared and administered?
A: Reconstitute the lyophilized powder with the recommended volume of sterile diluent (as guided by your healthcare professional) before intravenous infusion. Administer over the recommended period, following strict aseptic techniques. Each vial is for single use only.
Q: What are the main therapeutic uses of Ancolase-500?
A: Ancolase-500 is used for the treatment of serious Gram-positive bacterial infections, including MRSA, C. difficile colitis, endocarditis, and bacteremia. It is especially useful when precise dosing is needed or in patients with renal impairment or fluid restrictions.
Q: When is it appropriate to use the 500 mg dosage instead of higher strengths?
A: The 500 mg dosage is ideal for pediatric patients, those with renal impairment requiring lower doses, step-down therapy, or situations where minimizing infusion volume is critical, providing safer and more accurate dosing flexibility.
Q: Where should Ancolase-500 be stored before use?
A: Store Ancolase-500 vials in a cool and dry place, away from moisture and direct sunlight, to ensure product stability and maintain effectiveness throughout its 24-month shelf life.
Q: What is the process for obtaining Ancolase-500 in India?
A: Ancolase-500 is distributed, manufactured, and supplied in India. Healthcare organizations and pharmacies can source it through authorized pharmaceutical distributors or directly from the manufacturer, following standard procurement protocols.
Q: What are the key benefits of using Ancolase-500 compared to other strengths?
A: The 500 mg vial offers more precise dosing options, particularly beneficial for dose adjustment in renal impairment, pediatric care, and fluid-restricted patients. Its compatibility for step-down therapy provides additional treatment flexibility.