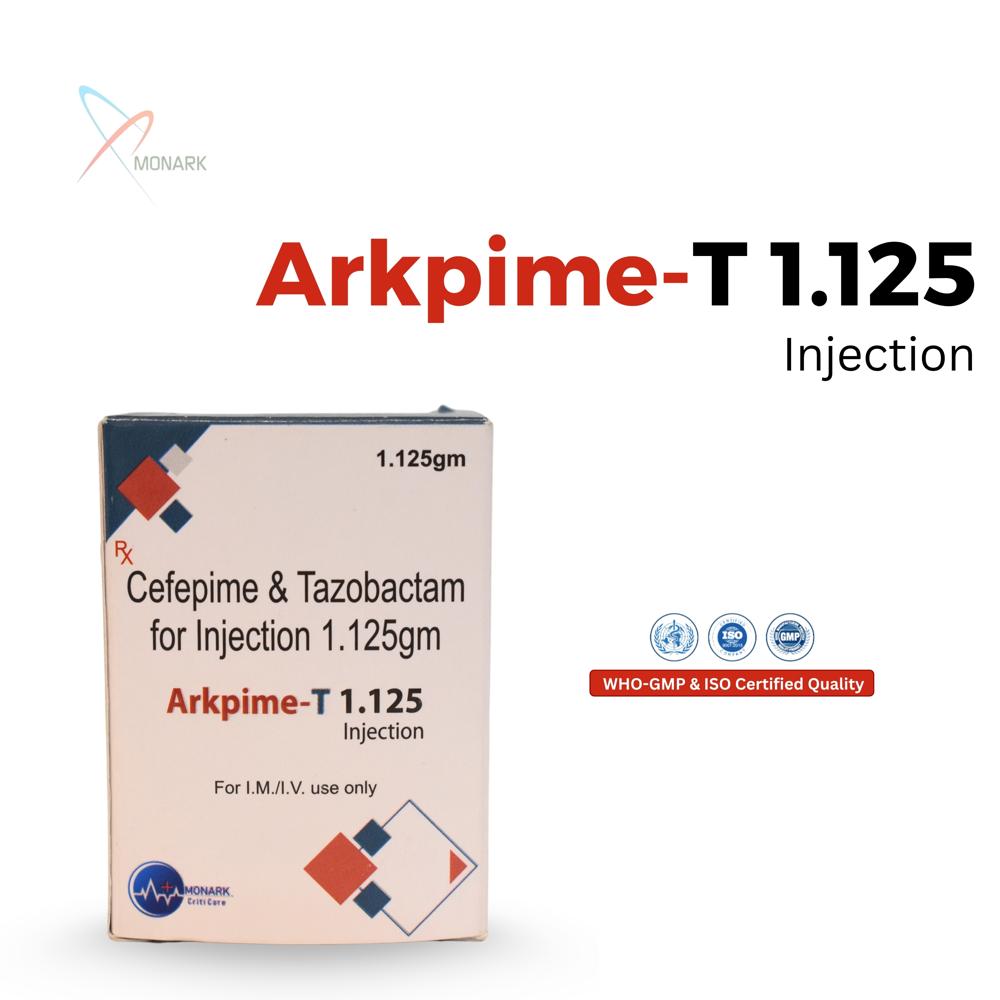
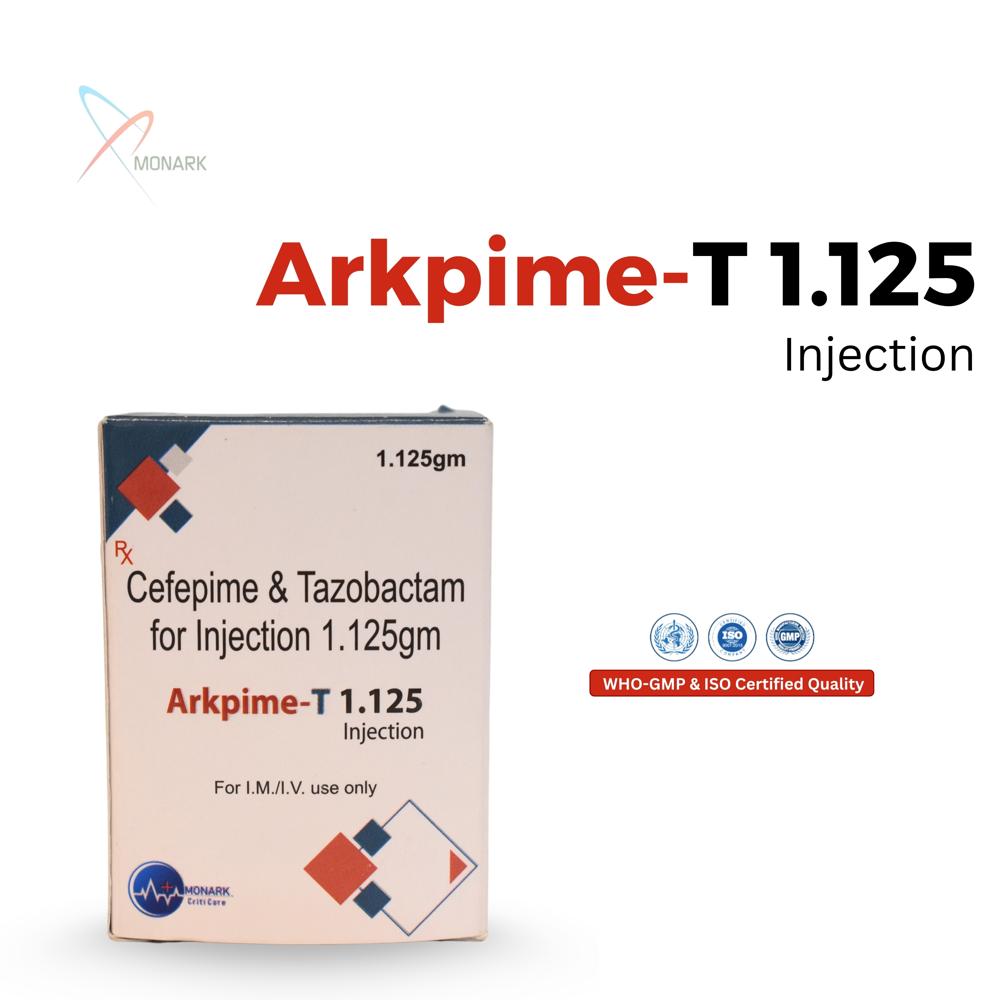
About Cefepime Hydrochloride 1000mg +Tazobactum Sodium 125MG INJ
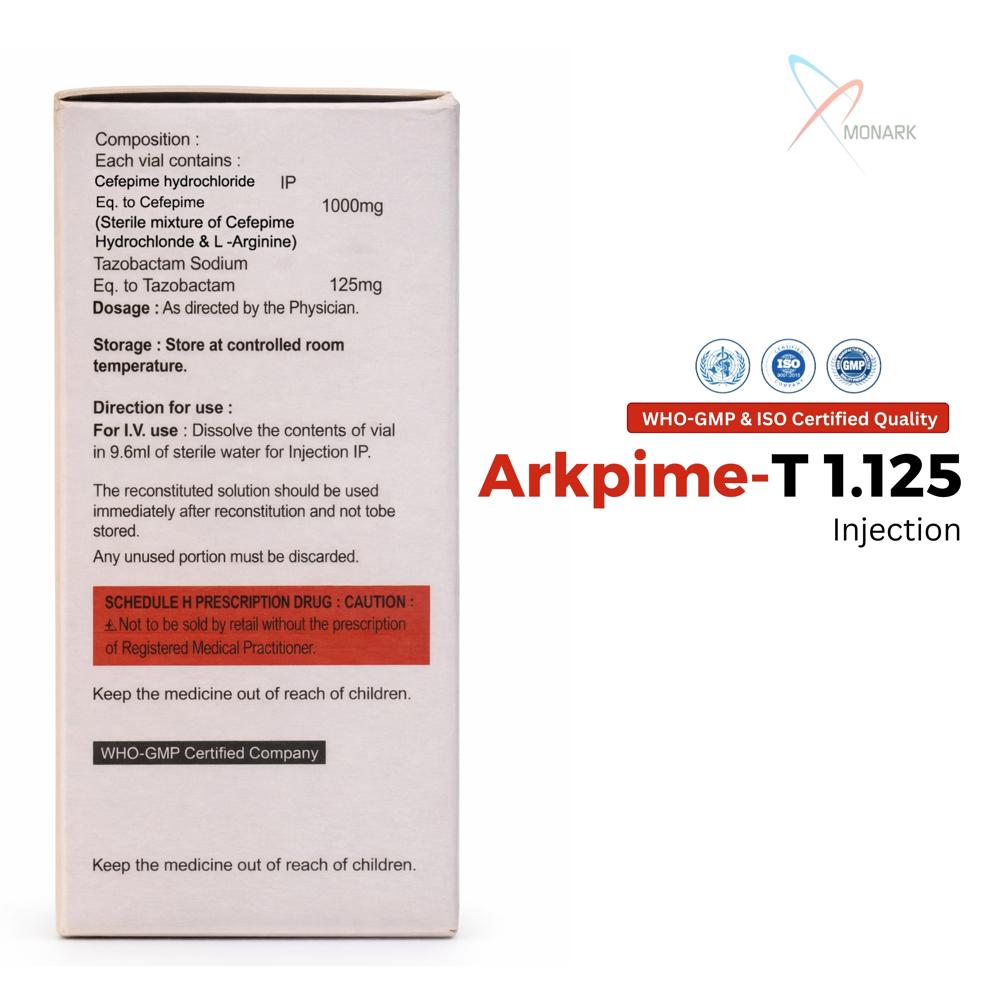
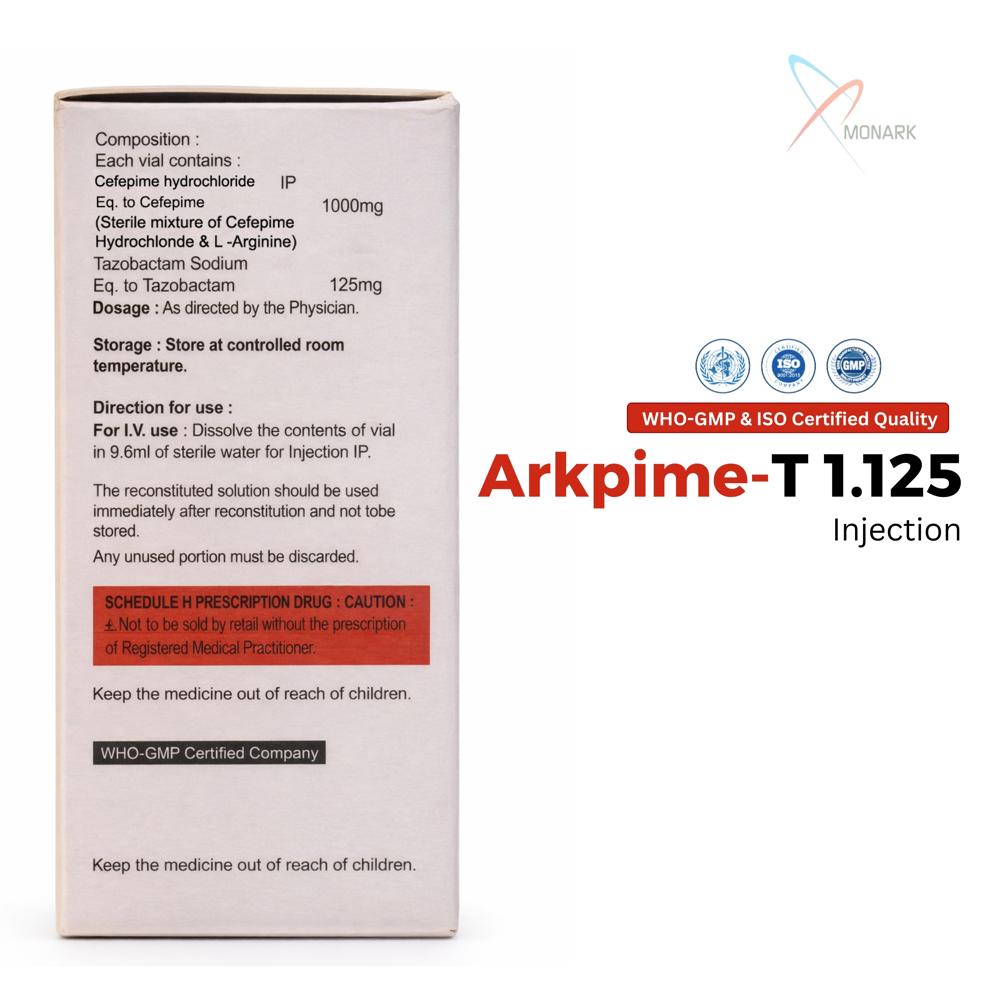
Cefepime Hydrochloride 1000 mg + Tazobactam Sodium 125 mg Injection is a sterile powder vial combining fourth-generation cephalosporin cefepime (1g) with beta-lactamase inhibitor tazobactam (125 mg) for IV/IM administration.
Powerful Combination for Broad Spectrum InfectionsArkpime-T 1.125 brings together Cefepime and Tazobactam, working synergistically to overcome beta-lactam resistant pathogens. This dual-action formulation makes it highly effective even against challenging Gram-negative and Gram-positive infections, providing robust coverage for healthcare professionals managing complicated bacterial diseases.
Flexible Administration and Convenient PackagingEach pack contains a single-use sterile vial, ensuring accurate dosing for hospital or clinical environments. Arkpime-T 1.125 can be administered by either intravenous or intramuscular routes, adapting to patient needs and severity of infection. The vial form ensures quick preparation and minimal waste.
Safe Storage and Long Shelf LifeArkpime-T 1.125 should be stored in a cool, dry place away from direct sunlight, preserving its efficacy for up to 24 months. Proper storage ensures the product remains sterile and effective for emergency or routine clinical use.
FAQ's of Cefepime Hydrochloride 1000mg +Tazobactum Sodium 125MG INJ:
Q: How does Arkpime-T 1.125 Injection work against infections?
A: Arkpime-T 1.125 combines cefepime, a cephalosporin antibiotic that inhibits bacterial cell wall synthesis, with tazobactam, a beta-lactamase inhibitor that protects cefepime from bacterial resistance. This combination allows it to treat a wide range of infections caused by both Gram-negative and Gram-positive bacteria.
Q: What infections can Arkpime-T 1.125 be used to treat?
A: This injection is indicated for complicated urinary tract infections, intra-abdominal infections, hospital-acquired pneumonia, skin and soft tissue infections, and sepsis. It is especially suitable where resistant organisms are a concern.
Q: When and how should Arkpime-T 1.125 Injection be administered?
A: The injection is administered either intravenously or intramuscularly by a healthcare professional, depending on the severity and type of infection. Dosage schedules should be determined by the doctor based on the patient's condition.
Q: Where should Arkpime-T 1.125 be stored for best results?
A: Store each vial in a cool, dry place away from direct light and moisture. Proper storage maintains the sterility and potency of the medication for its full 24-month shelf life.
Q: What is the benefit of combining cefepime with tazobactam in Arkpime-T 1.125?
A: Tazobactam enhances cefepime's effectiveness by inhibiting beta-lactamase enzymes that bacteria produce to resist antibiotics. This synergy provides improved efficacy against difficult-to-treat or resistant bacterial strains.
Q: Who manufactures and supplies Arkpime-T 1.125 Injection in India?
A: The medication is distributed, manufactured, and supplied by reputable pharmaceutical companies across India, ensuring quality and compliance with national regulatory standards.